December
22, 2005
Abbott
Receives FDA Approval for StarClose™ Vascular Closure
System
(source: Abbott)
An early holiday present for Abbott Vascular -- the FDA approved its StarClose
vascular closure device today -- a device that's been used successfully internationally
in over 55,000 patients. Abbott Vascular, which comprises a number of vascular
products, including the former market-leader in vascular closure devices, Perclose,
is now poised to compete with St. Jude Medical which markets Angio-Seal, the
leading closure device which utilizes a bioabsorbable suture and collagen plug
to form a seal on the artery. StarClose is quite different from the other devices
currently used, since it is a Nitinol clip which is applied on the outside
of the artery. The company claims that in this way, nothing remains inside
the artery to cause clotting or other complications. This also avoids any allergies
that patients may have to collagen (see our very
active Forum topic on vascular closure devices to read about the many
complications patients have experienced with the devices used to date). The
reason for using a vascular closure device instead of manual compression is
two-fold: one, the patient can sit up and walk much sooner; two, the femoral
artery can be closed more securely with less complications. The first has been
borne out, but currently the complication rates between manual and device-based
closure are not different, somewhere around 3%. It will be interesting to see
if Abbott's device can reduce this complication rate.
December
12, 2005
Patients
trust docs, but often use the Internet
(source: Reuters Health)
Patients in the United States are likely to first turn to the World Wide Web
to find out health information, according to a study published in the current
Archives of Internal Medicine that looked at patients looking for information
about cancer. Only about 11% said they actually consult their physician first,
but the study also showed that patients trusted the information from their
doctors more. The implications of this study are important because as lead
investigator Dr. Bradford W. Hesse pointed out, "Ongoing attention may be needed
to adjust reimbursement policies for time spent with patients interpreting
printouts, for accommodating shifts toward informed and shared decision making,
for steering consumers to credible information sources, and for attending to
the needs of those who fall through the cracks of the digital divide...."
related stories:
Trust
and Sources of Health Information -- Archives
of Internal Medicine
December
11, 2005
Cook's
industry is on the move
(source: Jeff Swiatek, Indianapolis Star)
A backgrounder on privately-held Cook Group, in the context of the acquisition
and merger news of the past week. The article relates how a few years ago the
company almost was bought out by Guidant, now "the" target of acquisition.
Cook is moving forward on several fronts: it currently is enrolling patients
in a trial of its drug-coated Zilver stent to see if it provides a solution
for blocked arteries in the leg. The company also has several versions of endografts,
used to repair aortic aneurysms.
December
8, 2005
Boston
Scientific Announces Worldwide Recall of Flextome Cutting
Balloon® Systems
(source: Boston Scientific Corporation)
Less than 3 months after receiving FDA approval for
its Flextome cutting balloon system, and 3 days after offering to buy Guidant,
Boston Scientific has issued a worldwide recall for the Monorail version of
the device. 40,000 cutting balloons are being recalled after the company received
8 complaints of problems where the distal (far) end of the device separated
from the delivery catheter as it was being withdrawn. This resulted in 3 patients
having to undergo surgery. No deaths were reported.According to the Wall Street
Journal, this was the 6th time this year that the company has had to take product
off the shelves.
related stories:
Boston
Scientific To Recall Catheters After 8 Complaints -- Wall
Street Journal $$
Boston
Scientific recalls artery devices -- Reuters
Boston
Wants Its Balloons Back -- Red Herring
December
7, 2005
Guidant
says it'll talk with Boston Sci about a deal
(source: Star Tribune, Minneapolis)
It's kind of "Well, I've already been asked to the prom by someone, but
now that you've asked me too, I'll think about it". Guidant agrees to
consider Boston Scientific's bid and allow Boston to do "due diligence",
which means Boston gets to look at the books. J&J is not happy, but feels
that their lower revised bid is still in the best interests of Guidant.
related stories:
For
now, J&J decides against a counteroffer to win Guidant -- Boston
Globe
Guidant,
new suitor plan talks -- Jeff Swiatek,
IndyStar
December
5, 2005
Boston
Scientific Proposes to Acquire Guidant for $72 Per Share
in Cash and Stock; Transaction Valued at $25 Billion
(source: Boston Scientific Corporation)
This is breaking news. In a surprise move, Boston Scientific has offered to
buy Guidant, besting Johnson & Johnson's bid by some $3 billion J&J's
bid was reduced from its original amount in response to problems Guidant had
over faulty defibrillators and the two companies agreed to
the reduced amount less than three weeks ago. Boston Scientific states that
they have financing agreements in hand. Of course, there is the FTC -- they
would need to start the review process of the merger all over again -- there
are a number of areas where Boston would need to divest itself of products.
Boston Scientific's CEO Jim Tobin said that the regulatory issues have been
reviewed and the company is prepared to make the required divestitures, mostly
the endovascular (non-cardiac) part of the business. He believes this acquisition
would be better for Guidant employees (he calls them "good people")
than J&J's offer. The combination of companies would be the third largest
medical device company in the world and would combine Guidant's cardiac rhythm
management (CRM) business, second only to Medtronic, with Boston DES and other
interventional products. CFO Larry Best said he believes the agreement could
be finalized by year-end with shareholder votes in the first quarter of 2006.
Stay tuned.
related stories:
Boston
Scientific Offers $25 Billion to Buy Guidant -- Associated
Press
Boston
Scientific Bids $25 Billion For Guidant, Trumping J&J -- Wall
Street Journal $$
November
29, 2005
Cook
Launches New Angioplasty Balloon Catheter With Advanced
Features
(source: Cook Incorporated)
Cook is launching the Advance ATB® PTA Dilatation Catheter, a new peripheral
balloon angioplasty catheter, optimized for ease of insertion and pullback
into the sheath. The Advance balloon incorporates an increased lumen size for
faster inflation/deflation rates while still providing sufficient rated burst
pressure to dilate demanding lesions.
November
29, 2005
Hospital
gets new intravascular imaging system
(source: Dallas-Fort Worth Star Telegram)
A local news story that shows how new imaging techniques like Volcano Therapeutics'
colorized intravascular ultrasound (IVUS) and 64 slice multislice computed
tomography (MSCT) are changing the way coronary artery disease is diagnosed.
November
29, 2005
Emergency
bypass surgery on angioplasty patients drops 90 percent
(source: American College of Cardiology)
The experience of Mayo Clinic is recounted in this study appearing in the current
issue of Journal of the American College of Cardiology -- the data is in, confirming
what everyone has known: that stents have radically reduced the need for emergency
bypass surgery to salvage an angioplasty gone bad. In the words of Dr. Mandeep
Singh, "We knew there had been a reduction, but the magnitude of the reduction
was a surprise to us.... The bypass surgery rates, which were close to 3 percent,
came down to 0.3 percent in the most recent time period."
related stories:
Emergency
Coronary Artery Bypass Surgery for Percutaneous Coronary Interventions -- Journal
of the American College of Cardiology
November
29, 2005
Stent
vs. Scalpel
(source: Barnaby J. Feder, New York Times)
An excellent overview of the controversy between CES and CEA (stenting and
surgery) discusses questions like which is better?; are both treatments being
over-used?; does opening up the carotid artery really prevent stroke?
related stories:
Interview with
Christopher J. White -- Angioplasty.Org
Conducted this spring, Dr. White, a cardiologist at the reknown
Ochsner Clinic in New Orleans, discusses indications for carotid stenting
and the current state of the art.
November 29, 2005
Boston
Scientific takes its show on the road
(source: Stephen Heuser, Boston Globe)
A report on Boston Scientific's simulation bus, the SimSuite -- actually two
articles and an audiovisual slideshow.
November
29, 2005
Little-known
heart therapy helps squeeze out chest pain
(source: Robyn Shelton, Orlando Sentinel)
An article from a Florida newspaper about EECP (Enhanced External Counterpulsation),
a non-invasive therapy to improve circulation that's been around for a while
and, amid skepticism, is being used to treat heart patients. The PEECH study,
presented at the ACC earlier this year, found that patients suffering from
congestive heart failure (CHF) had better exercise duration when treated with
EECP than with pharmacologic management alone.
related stories:
Vasomedical
Announces Positive PEECH Trial Results -- VasoMedical
Inc. (March 9 press release)
Forum
Discussion Topic on EECP -- Angioplasty.Org
November
17, 2005
Medtronic
Submits First PMA Module for Talent™ Thoracic Stent
Graft System
(source: Medtronic, Inc.)
From the company release: "Each year in America, thoracic aneurysms affect
approximately 30,000 people. Patients typically have no symptoms and, when
they are diagnosed, often undergo complex open surgical repair. Many of these
patients have other serious conditions such as coronary artery disease, high
blood pressure or diabetes that make surgical repair difficult or impossible.
For those patients, conservative medical management or "watchful waiting" is
sometimes selected as a treatment option. However, this can lead to increased
mortality and morbidity. Stent graft therapy provides a minimally-invasive
alternative, with reduced recovery times and potentially improved survival
rates."
November
15, 2005
Abbott
to Acquire License to Johnson & Johnson's Rapid Exchange
Catheter Delivery Technology
(source: Abbott)
As expected, Abbott gets the Rapid Exchange RX technology from the J&J/Guidant
deal, as a requirement made by the FTC. This now means that Boston Scientific,
J&J/Cordis/Guidant and now Abbott have the capability of making systems
with Rapid Exchange, leaving Medtronic as the lone non-RX device manufacturer.
November
15, 2005
Smoking
Ban Reduces Heart Attacks in Colorado City by 27%
(source: Helena MT Independent Record)
Backing up the results of a similar experiment in Helena, Montana, the community
of Pueblo, Colorado experienced 27% less hospital admissions for heart attacks
in the 18 months after a public smoking ban went into effect. The Montana law
was in effect only 6 months when a judge struck down the law. Heart attack
admissions went back up to previous levels soon after.
related stories:
Study:
Heart Attacks Drop With Smoking Ban -- Associated
Press
Heart
attacks pared in Pueblo -- Denver Post
November
15, 2005
Johnson & Johnson
and Guidant Corporation Announce Revised Acquisition Agreement
with Net Value of $19 Billion
(source: Johnson & Johnson; Guidant Corporation)
After a rocky couple weeks, the deal is back on -- at a $4 billion lower price
tag.
related stories:
Johnson
and Guidant Agree on Revised Buyout Deal -- New York
Times
J&J
to Pay $4 Billion Less For Guidant in Revised Deal -- Wall
Street Journal ($$)
J&J
reduces Guidant offer -- Reuters
November
13, 2005
Major
advances in techniques, devices, medications spur update
of PCI Guidelines
(source: American College of Cardiology)
Today the American College of Cardiology (ACC), American Heart Association
(AHA), and Society for Cardiovascular Angiography and Interventions (SCAI)
released the first major overhaul of professional guidelines for angioplasty
and stenting in four years. The guidelines take into account the drug-eluting
stent revolution, expanded applications for percutaneous coronary interventions
(PCI) and add to the growing body of practice-based medicine for treatment
of heart attacks, prescription of antiplatelet therapy post-PCI, etc. While
these guidelines tend to be conservative, of note is the recommended use of
drug-eluting stents as therapy for in-stent restenosis -- a use not yet approved
by the FDA for the stents currently available in the U.S. A summary
of the guideline updates can be found on the ACC website.
November
13, 2005
Stent
patients go home sooner if wrist artery used
(source: Reuters)
The Canadian study acronymed EASY (Early Discharge After Transradial Stenting
of Coronary Arteries Study), which was previously presented at the Paris Course
on Revascularization and the recent Canadian Cardiovascular Congress, was presented
at today's session of the American Heart Association Scientific Sessions 2005,
and so is finally receiving some press in the U.S.. The radial (wrist) approach
to threading a catheter into the coronary arteries has been around for a number
of years, but it requires special training and skill on the part of the cardiologist
(most catheter-based procedures currently utilize the femoral or groin artery
as an entry point). ReoPro, or similar drugs, are routinely given after any
stenting to reduce complications from blood-clotting -- usually the patient
is kept overnight on an IV drip of the drug. What this study showed was that
the overnight infusion was not necessary and added no benefit to a single dose
-- the implication is that patients can go home the same day, in effect transforming
angioplasty with stenting into an outpatient procedure. There is, however,
debate in the medical community as to whether patients should be kept overnight.
One of the big advantages of radial angioplasty is instant ambulation -- that
is, the patient can walk around shortly after the procedure and does not need
to lie flat or have a vascular closure device used, both of which occur in
groin artery cases.
related stories:
Angioplasty
Becoming Outpatient Procedure -- Associated Press
November
7, 2005
St.
Jude Medical Named Medical Manufacturer of the Year
(source: Medical Device & Diagnostic Industry)
The magazine has named three companies to this distinction, a large company,
a small public company and a small private company. St. Jude, maker of implantable
cardio defibrillators (ICD), Angio-Seal, heart valves and many other devices
is profiled in the story, "A Lot of Hard Work and Just a Little Luck:
St. Jude Medical proves that good processes and a good reputation are key ingredients
for success."
related stories:
St. Jude Medical
Named Medical Device Manufacturer of the Year -- St.
Jude Medical
November
7, 2005
Guidant
Initiates Suit for Specific Performance
(source: Guidant Corporation)
The Monday morning card is played by Guidant in the current medical device
acquisition poker game: Guidant, which is being sued
for fraud by NYS Attorney General Eliot Spitzer, is now suing J&J
to enforce their acquisition agreement. For their part, J&J
has stated that they have the right to renegotiate the terms at a lower price,
due to the various recall and legal problems that Guidant has experienced in
connection with their defibrillator products.
related stories:
Guidant
Sues to Force J&J to Complete Acquisition -- Bloomberg
News
Guidant
Sues Johnson & Johnson for Completion of Merger -- New
York Times
November
4, 2005
For
Americans, Getting Sick Has Its Price:
Survey Says U.S. Patients Pay More, Get Less Than Those in Other Western Nations
(source: Ron Stein, Washington Post)
Americans were the most likely to pay at least $1,000 in out-of-pocket expenses.
More than half went without needed care because of cost and more than one-third
endured mistakes and disorganized care when they did get treated.
November
4, 2005
COMMIT
Trial: Addition of clopidogrel to aspirin safely reduces
mortality in patients with acute myocardial infarction
(source: The Lancet)
The COMMIT Trial (ClOpidogrel and Metoprolol in Myocardial Infarction Trial)
randomized 45,852 patients to either clopidogrel (brand name -- Plavix) or
placebo within 24 hours of admission for acute myocardial infarction (heart
attack) at 1,250 hospitals in the study. The use of clopidogrel resulted in
a 9% reduction in death, reinfarction, or stroke with no significant excess
risk. A 75mg dosage of the drug was continued for up to 4 weeks. Clopidogrel
(Plavix) is a crucial post-procedure medication for stent patients (especially
drug-eluting stents) but the COMMIT study shows that its antiplatelet properties
have a clear therapeutic effect even when stents are not used.
related stories:
Cheap
drugs could slash heart attack death toll -- Guardian
Unlimited
A bit of currency exchange information is in order. "Cheap" in
England is £1.25/day (equal to $2.21 US) for Plavix; however,
in the United States, even through discount online sources such as Walgreens.com,
the drug costs almost twice as much ($4.20/day). Still, that's not
much more than a latte -- and it does more than wake you up.
Sanofi's
Plavix May Reduce Heart Attack Deaths, Study Shows -- Bloomberg
November
3, 2005
Guidant
Sued By Spitzer for Misleading Doctors
(source: New York State Office of the Attorney General)
Guidant's problems get worse as New York Attorney General Eliot Spitzer filed
a fraud lawsuit, stating that the company misled doctors about a design flaw
in a heart defibrillator. Spitzer said, "Concealment of negative facts that
might influence a consumer to purchase another manufacturer's product is the
essence of fraud."
related stories:
Troubled
Maker of Heart Devices May Lose Suitor -- New York
Times
Johnson & Johnson
Threatens To Pull Plug on Guidant Deal -- Wall
Street Journal
November
2, 2005
J&J
May Cancel $25.4B Guidant Acquisition
(source: Associated Press)
Johnson & Johnson Says It Might Not Complete Guidant Acquisition Despite
FTC Clearance because of problems with ICDs, defibrillators and recalls since
the deal was announced. Some think J&J is bargaining for a lower price;
some think they may just want out; Guidant's stand is that J&J is obligated
contractually to go through with the deal; J&J thinks otherwise (read their
press releases below):
Federal
Trade Commission Clears Johnson & Johnson's Acquisition of Guidant -- Guidant
Corporation
Johnson & Johnson
Receives FTC Clearance of Proposed Acquisition of
Guidant Corporation and Provides Update on Transaction in Light of Guidant
Recall Events -- Johnson & Johnson
related stories:
J&J
says may not close Guidant deal -- Reuters
Johnson
Threatens to Drop Plan to Buy Guidant -- New
York Times
If
Not Guidant, Then Who? -- Forbes.com
J&J
Casts Doubt On Deal for Guidant -- Wall
Street Journal
November
1, 2005
U.S.
Approval Is Expected Soon for Sale of Guidant
(source: New York Times)
Speculation about whether or not Johnson & Johnson will go through with
acquiring Guidant or, if they do, how much lower the offer will be will soon
be over. Once the FTC approves the deal, the companies have 48 hours to sort
things out. The price offered may be lowered due to the recent revelations
about problems with Guidant defibrillators.
related stories:
FTC
could OK J&J-Guidant Tuesday -- Reuters
October
25, 2005
Cook
Offers First Fenestrated Endograft With CE Mark Approval
For Complex Aortic Aneurysms
(source: Cook Incorporated)
The company announced that its advanced AAA endograft has received the European
CE Mark approval and that a pilot study is now ongoing in the United States.
This device allows endovascular (catheter-based) repair of Abdominal Aortic
Aneurysms.
October
20, 2005
Repeated
Defect in Heart Devices Exposes a History of Problems
(source: Barry Meier, New York Times)
A major story discussing the defects in Guidant's defibrillators, and the roles
of the company, doctors, patients and the FDA. While not a topic involving
interventional cardiology, stents, balloons or angioplasty, this article is
about the world of medical devices, recalls and how they impact the health
of patients. Guidant's press release responding to the NYT article is posted
below.
related stories:
Guidant
Response to Media Story on Product Performance -- Guidant
Corporation
Guidant
may have leverage in talks with J&J -- Reuters
 October
20, 2005 October
20, 2005
Guidant
Announces Positive Preliminary Results of Large Real-World
Carotid Artery Stenting Study
(source: Guidant Corporation)
The company announced preliminary results of its study of carotid artery stenting
in high-risk patients, called CAPTURE (Carotid ACCULINK/ACCUNET Post Approval
Trial to Uncover Rare Events) -- an FDA-required post-approval study to determine
whether carotid artery stenting can be performed safely in real-world clinical
settings with physicians of varying levels of experience. Initial results confirmed
positive earlier data on carotid stenting for high-risk patients. In addition,
the study suggests that carotid artery stenting can be performed safely by
physicians of different experience levels. The rate of death, stroke and myocardial
infarction within 30 days of the procedure was 5.1 percent.
 October
17, 2005 October
17, 2005
Boston
Scientific Study Shows Excellent Performance in High-Risk
Patients with Carotid Artery Disease
The BEACH trial demonstrated that Boston Scientific's Carotid WALLSTENT® Monorail® Endoprosthesis,
used in combination with the Company's FilterWire EX® and FilterWire EZ™ Embolic
Protection Systems, improved blood flow within the vessel and the vessel remained
open up to one year post-procedure in patients at high risk for surgery (carotid
endarterectomy).
(source: Boston Scientific Corporation)
related stories:
Report
on Stents Could Spur Increase in Medicare Aid -- New
York Times
 October
17, 2005 October
17, 2005
Continued
Improvement and Outstanding Safety Seen with Boston Scientific's
FilterWire EZ™ Embolic Protection System
In about a third of surgical coronary bypass patients, saphenous vein grafts
(SVG) develop clogging; these clogs are then reopened using the less invasive
procedure of angioplasty. A problem has been that the interior arterial wall
and plaque in these SVGs are more prone to embolization, where small pieces
of the plaque break off and travel through the bloodstream into smaller arteries,
often causing Major Adverse Cardiac Events (MACE, i.e. heart attack, etc.).
To prevent this, embolic protection devices are used to filter and catch the
plaque and prevent them from traveling onward. Still, the MACE rate is abolut
10%. Two registries presented by Boston Scientific show MACE rates reduced
to about 5% using their FilterWireEZ (not yet approved in the U.S.).
(source: Boston Scientific Corporation)
 October
17, 2005 October
17, 2005
Volcano
Corporation Announces Pioneering Intravascular Ultrasound
(IVUS) Vulnerable Plaque Study
(source: Volcano Corporation)
The SPECIAL (Study of Prospective Events in Coronary Intermediate Atherosclerotic
Lesions) trial is in collaboration with Goodman, Fukuda Denshi, Toyohashi Heart
Center, and will look at up to 2,000 stent patients in 100 hospitals in Japan
to study the natural history of the disease process of atherosclerosis. While
it has been thought for some time that heart attacks occurs when the artery
becomes blocked over time, it is now suggested that these events may be more
acute and related to the type of plaque -- a vulnerable lipid-rich plaque that
has not caused symptoms of angina may rupture, block the artery, causing an
infarction of the heart. The Volcano intravascular ultrasound IVUS system can
characterize not only the amount of plaque but its tissue characteristics.
Pauliina Margolis, M.D. PhD, Medical Director for Volcano and study collaborator
states, "The SPECIAL study will collect critical new information about the
role of vulnerable plaque in unexpected heart attacks and about the natural
progression of coronary artery disease.... As a growing body of scientific
knowledge about vulnerable plaque is built, physicians may one day be able
to better predict and prevent heart attacks and plaque progression."
 October
17, 2005 October
17, 2005
GE
Healthcare and Volcano Corporation Collaborate to Develop
a State-of-the-Art Cath Lab System with Fully Integrated
IVUS Capabilities
(source: Volcano Corporation)
This collaboration agreement is intended to bring an industry-first imaging
capability to cardiac catheterization labs around the world. Under terms of
the agreement, GE and Volcano will collaborate to develop a state-of-the-art
digital cardiovascular imaging system with fully integrated intravascular ultrasound
imaging (IVUS) capabilities.
October
4, 2005
New
procedure cuts risk in Iowan's heart surgery
(source: DesMoines Register)
One of the ironies of heart disease is that the patient who most needs a coronary
intervention (for example, a stent) may have a heart that is too weak to have
the procedure performed safely. The chances of a serious complication or death
during an interventional procedure or surgery in this patient population is
high. Should such a "high risk" procedure have to be done, a device
called an Intra Aortic Balloon Pump (IABP) is often used to help support the
pumping of the heart. But sometimes it doesn't provide enough support, or can't
be used. Another procedure we've reported on called Percutaneous Cardiopulmonary
Bypass Support (PCPS) also addresses this problem (see
our interview with Dr. Fayaz Shawl) -- it is in effect a bypass
that is done in the cath lab without open heart surgery. But it takes significant
training to learn and time to perform. Two years ago the FDA approved a device
called the TandemHeart®, made by Pennsylvania-based CardiacAssist.
It is a small device that can be inserted percutaneously through the same puncture
in the groin that is used for angioplasty and stenting. It takes about an hour
to insert into the heart and certainly takes special training to use but, as
this article describes, it can provide enough pumping power so that angioplasty/stenting
(or other intervention) can be performed much more safely in a "high risk" patient.
September
29, 2005
Trial
Will Look At Widening Angioplasty's Reach
Cardiologists at Johns Hopkins have launched a nationwide study of more than
16,000 patients to see if angioplasty can be safely performed in smaller, community
hospitals, easing access to the therapy for patients. Researchers expect to
enroll the first study patients in this fall. This controversial topic was
the subject of one of the earliest
postings on our Forum -- by Johns Hopkins interventional cardiologist
Thomas Aversano, M.D. who is leading the above study.
(source: Robert Preidt, HealthDay)
September 28, 2005, 2006
Cordis
Corporation Acquires LuMend, Inc.
(source: Cordis Corporation)
Cordis Corporation, a Johnson & Johnson company, today announced the acquisition
of LuMend, Inc., a privately held company focused on the development of chronic
total occlusion (CTO) devices to treat peripheral vascular disease.
September
16, 2005
Boston
Scientific Receives FDA Clearance for Flextome™ Cutting
Balloon® Dilatation Device
(source: Boston Scientific Corporation)
The FDA has now approved this improved version of an earlier Cutting Balloon
-- this one is more "deliverable". The problem it addresses is that
during angioplasty the plaque itself splits apart when the balloon expands.
In a complex lesion, this splitting may be quite eccentric and unpredictable;
sometimes difficult and not possible even under high pressures. Sometimes hard
plaque can dissect or rip the arterial wall. This new device uses tiny longitudinal
blades to slice into the plaque before expansion, making the angioplasty easier,
more predictable and successful.
September
15, 2005
Abbott
Receives U.S. Approval for New Device Designed to Treat
Patients at Risk of Stroke
(source: Abbott)
Abbott now becomes the second company to market a carotid stent/embolic protection
device system in the U.S. Guidant's AccuLink/AccuNet system was approved last
August. For more information on carotid stenting, read our
interview with Dr. Chris White.
July
25, 2005
Carotid
therapy studied at Christ Hospital (Cincinnati)
Report on an NIH study being conducted, comparing surgery vs. carotid stenting
in low surgical risk patients. The stenting procedure is currently recommended
only for high surgical risk candidates.
(source: Roy Wood, Cincinnati Post)
July 19,
2005
Magical
Medicine on TV
Dr. Jauhar provides a review of the TV show "House". Read more comments
in our Editor's
Blog.
(source: Sandeep Jauhar, M.D., New York Times)
July
15, 2005
More
Americans Go Online For Health-Care Information
The actual number of adults who have ever searched for health information online
has grown due to an increase in the percentage of U.S. adults who go online
(from 66% in 2002 to 74% in 2005). Approximately 117 million adults have searched
for health information in the past year, and almost six in 10 (58%) say that
they have looked for information about health topics often (25%) or sometimes
(33%), an increase of eight percentage points from 2004 (50%).
(source: Harris Interactive)
July 11,
2005
Paswan's
brother for CBI probe into Escorts' medical advice
A story from New Delhi, India, about bypass surgery vs. angioplasty in the
case of MP and Union Minister Ram Vilas Paswan -- turns out the original hospital
he went to recommended immediate surgery without question -- but a noted cardiologist
from a different hospital said angioplasty was indicated. Minister Paswan was
transferred, had the angioplasty, is at work from his hospital room and is
expected to be discharged in a day or two.
(source: The Hindu)
July 5,
2005
For
Heart Disease, a Joint Effort
A report about a hybrid procedure offered in Maryland which combines coronary
artery bypass grafting (CABG) with Angioplasty/Stenting in a single procedure,
using the philosophy that both therapies work for different types of cases.
The procedure is not without controversy.
(source: Naseem Sowti, Washington Post)
July 4, 2005
Cubin
hospitalized for mild heart attack
Rep. Barbara Cubin (R-Wyo) suffered a mild heart attack at Dulles International
Airport and was taken to a Washington-area hospital where an angioplasty/stent
was done. Her spokesman said she was in good spirits and "joked that she's
always trying to follow in Dick Cheney's footsteps but this was going too far.''
(V.P. Cheney has had four heart attacks.)
(source: Caspar Star Tribune)
June
28, 2005
Recalls
cast spotlight on medical device sector
The latest recall from Guidant is discussed in the context of other high profile
device recalls over the past year. Recalls are a complex area -- read our feature "What's
a Patient To Do" which was specifically about the Boston Scientific
stent recall of last summer, but has information about devices recalls in general.
(source: Susan Kelly, Reuters)
July 4, 2005
The
Frontier: Interventional Radiology
A feature in the July 4 issue of Newsweek profiles the field of IR, Interventional
Radiology, and the advances being made in catheter-based treatments outside
of the heart.
(source: Robert Bazell, Newsweek)
June
22, 2005
Centers
for Medicare and Medicaid Services Initiates Review for
Expanded Reimbursement Coverage of Vasomedical's EECP® Therapy
(source: Vasomedical, Inc.)
The company has been notified by the Centers for Medicare and Medicaid Services
(CMS) that the application requesting consideration for expansion of the national
coverage for EECP® therapy has been accepted. The company currently markets
the therapy for chronic stable angina and congestive heart failure and has
been actively working to expand Medicare reimbursement coverage for less severe
angina and to include coverage for congestive heart failure as a primary indication.
June 21, 2005
Heart
Rhythm Society to Develop Guidelines on ICD Recalls
In reaction to the recent recalls from Guidant, and earlier from Medtronic,
the Heart Rhythm Society is calling together a task force to ensure that recall
information in implantable devices (defibrillators, pacemakers, etc.) is reported
to both physicians and patients in a standardized way.
(source: Heart Rhythm Society)
June
20, 2005
Recall
of certain Guidant implantable heart devices
The AHA's statement on Guidant's recall of defibrillators. The models are PRIZM
2 DR, Model 1861, manufactured on or before April 16, 2002, CONTAK RENEWAL,
Model H135, manufactured on or before August 26, 2004, CONTAK RENEWAL 2, Model
H155, manufactured on or before August 26, 2004
(source: American Heart Association)
June
14, 2005
Acute
coronary syndrome patients at increased risk
of stroke
A study published in the current Circulation shows that patients admitted for
acute heart problems have a higher rate of stroke, both in the hospital and
after discharge -- this confirms that what medicine knows but need to pay far
more attention to -- vascular disease occurs in different parts of the body
-- in the coronary arteries it causes heart attacks; in the carotids it causes
strokes; in the renals, it causes kidney disease; in the legs, PAD -- but these
seeming different manifestations are all related and often occur together.
(source: American Heart Association)
June 2, 2005
Palestinian
Leader Has Heart Procedure
Palestinian leader Mahmoud Abbas is recovering from angioplasty performed Wednesday
at a hospital in Jordan. Abbas was taken to a hospital in Amman, Jordan Wednesday
complaining of fatigue, said Tayeb Abdel Rahim, a top Abbas aide. He underwent
angioplasty, a procedure to clear out clogged coronary arteries.
(source: Mohammed Daraghmeh, Guardian Unlimited)
May
30, 2005
Statins
underprescribed to patients at higher risk of heart disease
A study published in the May issue of the Public Library of Science-Medicine by
a Stanford team, headed by lead author Jun Ma, MD, PhD, found that statins,
although their benefit in lowering cholesterol is well-known, are still not
being prescribed for the patients who need the therapy most. Dr. Ma stated
that while she was encouraged that statin use has increased, “it’s
disconcerting that the magnitude of the increase is much smaller than expected.
The rate of use falls significantly short of the latest recommendations.” Ma
also noted that although statin use is greatest among the high-risk group,
physicians should be more aggressive in looking at statin therapy for moderate-risk
patients. “Prevention is key for delaying the onset of cardiovascular
disease. If we start intervention early enough, we can delay the progression
of the disease and reduce the health-care costs overall.”
(source: Stanford University Medical Center)
May 25,
2005
Noninvasive
Coronary Angiography With Multislice Computed
Tomography
MSCT is a technology that has been talked about as an alternative to cardiac
catheterization. It is not catheter-based and therefore avoids the complications
of percutaneous procedures (about 1.8%). Today's study in JAMA reports on 103
patients in Germany who underwent both invasive coronary angiography and MSCT
(using a scanner with 16 detector rows manufactured by Philips Medical Systems).
The authors from University Hospital in Ulm, Germany, conclude that MSCT is
effective in pre-screening patients for coronary angiography. For example,
45 patients showed no or insignificant coronary disease on their angiogram,
currently the "gold standard" for diagnosis. However, the less invasive
MSCT identified 38 of these patients correctly -- that means 84% of these patients
could have avoided getting a cardiac catheterization. There were only 2 patients
who had coronary artery narrowing that were missed by the MSCT and it is thought
that with the current state-of-the-art technology (40 and 64 detector rows)
these too may have been found. The authors conclude: "With rapidly improving
technology, MSCT may well evolve from a useful complement to invasive angiography
to a clinically viable alternative." This would truly be a revolution
in imaging for patients (reduced complications) and healthcare systems (reduced
costs).
(source: Journal of the American Medical Association)
related stories:
Multislice
Computed Tomography Has Potential To Offer Accurate,
Safer Method for Detecting Coronary Artery Disease -- Journal
of the American Medical Association
May
19, 2005
Relation
of Interhospital Delay and Mortality in Patients
With ST-Segment Elevation Myocardial Infarction
Transferred for Primary Coronary Angioplasty
Published online today, this study confirms the fact that that longer the
delay in treating STEMI (ST elevated myocardial infarction) patients with
angioplasty / stenting, the worse the outcome. As can be seen in this graph,
1-year mortality increased from 3.2% to 12.1% as the time delay due to
interhospital transfer increased from less than 30 minutes to greater than
90 minutes. Moral of the story: these times need to be reduced. Patients
with treatable MI should try to get to a hospital that can perform angioplasty.
(source: American Journal of Cardiology) |
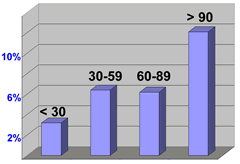
Time delay in
min. vs. % mortality at 1-year |
May
17, 2005
Market
for neck artery stents set to heat up
Carotid stents and stenting, a balloon/stent alternative to open surgery to
clean out the carotid artery is starting to become a force in the device industry.
For more on this new procedure, read our recent
interview with Dr. Christopher J. White of the Ochsner Clinic. A feature
story on the state of carotid stenting will be posted on Angioplasty.Org next
week. Register for our newsletter
to be notified.
(source: Susan Kelly, Reuters)
May
16, 2005
Class
Matters: Heart Attacks, and What Came Next --
Life at the Top in America Isn't Just Better,
It's Longer
This excellent article (part of a special New York Times series, "Class
Matters") profiles three New Yorkers who had heart attacks. The article
traces their treatments and outcomes, against the backdrop of their economic
class. The most successful of the three treatments was -- emergency angioplasty.
The article states, "An important link in the safety net that caught Mr.
Miele was his wife.... While Mr. Miele was still in the hospital, she was on
the Internet, Googling stents." We'd like to think that she read our article, "Angioplasty
and Heart Attack".
(source: Janny Scott, New York Times)
April 27,
2005
Boston
Scientific Announces Excellent Results in Study
of Patients With Carotid Artery Disease
The one-year data from the BEACH carotid artery stenting (CAS) clinical trial
were presented today at the 14th
Annual Peripheral Angioplasty and All That Jazz meeting in New Orleans
by Christopher White, M.D., of the Ochsner Heart and Vascular Institute in
New Orleans. The BEACH trial was designed to evaluate Boston Scientific's Carotid
WALLSTENT® Monorail® Endoprosthesis (Carotid WALLSTENT) and the FilterWire
EX™ and EZ™ Embolic Protection Systems and evaluated the effectiveness
of stenting with embolic protection for patients who are at high-risk for the
traditional surgical treatment of carotid endarterectomy (CEA). The combined
endpoint of stroke, death or heart attack at one-year with the stent was 9.1%,
lower than the results from the SAPPHIRE trial reported in October 2004 (that
trial showed stenting with a 12.2% and open surgery with a 20.1% mortality
or morbidity in the first year). The study included a subset of patients with
carotid disease in both left and right arteries, a population considered harder
to treat and one that has not been previously studied. This group showed an
event rate of 7.1%.
(source: Boston Scientific Corporation)
April 27, 2005
Guidant
Shareholders Overwhelmingly Approve Acquisition
Agreement with Johnson & Johnson
The merger is anticipated for the third quarter of this year, although regulatory
review in the European Union and U.S. is still pending.
(source: Guidant Corporation)
April
25, 2005
A
Model Patient: How simulators are changing the
way doctors are trained
A most interesting article about how doctors learn to do endovascular procedures
-- including carotid stenting.
(source: Jerome Groopman, New Yorker Magazine)
April
22, 2005
Abbott
Reviews 2004 Accomplishments at Annual Shareholders' Meeting
A press release from Abbott, a company which has begun drug-eluting stent trials
for its Zomaxx™ stent in Europe and also recently submitted its new StarClose® vascular
closure device for FDA approval. The company's Xact® Carotid
Stent System and Emboshield® Embolic Protection System for carotid
stenting is also currently under FDA review.
(source: Abbott Laboratories)
April 21, 2005
Guidant
Reports Record First Quarter Sales of $953 Million
The company's report contains some statements about the upcoming merger and,
a sign of the change in the field of angioplasty / stenting, stent sales (Guidant
only sells bare metal stents) were down 33%. This, of course, will change in
the near future, when the Guidant/J&J merger occurs.
(source: Guidant Corporation)
April
19, 2005
Boston
Scientific Announces First Quarter Results
The company announced financial results showing $721
million in stent sales worldwide. In a conference call with analysts
this morning, COO Paul LaViolette stated that the Taxus stent currently has
61% of the U.S. market.
(source: Boston Scientific Corporation)
April 19, 2005
Johnson & Johnson
- The World's Most Broadly Based Health Care
Company Reports 2005 First-Quarter EPS Rose 16.9%
on Sales Increase of 11.0%
In its press release, the company stated that its medical device division increased
sales over 13% to $4.8 billion. The report did not break out stent sales.
(source: Johnson & Johnson)
April
18, 2005
Boston
Scientific Announces Acquisition of TriVascular,
Inc.
Boston
Scientific Announces Decision to Acquire CryoVascular
Systems
The company announces acquisition of two device companies.
(source: Boston Scientific Corporation)
April
14, 2005
Boston
Scientific and Rubicon Medical Announce CE Mark
for the Rubicon Embolic Protection Filter
A new embolic protection filter that traps and removes debris that may be dislodged
during interventional procedures, has been approved for sale in Europe. In
a related story, Boston Scientific is exercising its option to purchase
the part of Rubicon it does not now own.
(source: Boston Scientific Corporation / Rubicon Medical)
April
13, 2005
EndoTex
Interventional Systems Announces European Approval
of Novel Carotid Stent Delivery System
Boston
Scientific Begins Exclusive International Distribution
of NexStent™ Carotid Stent
Both companies announce that Endotex's NexStent™ Carotid Stent and Monorail™ Delivery
System has received the CE Mark of approval in Europe and that Boston Scientific
will begin exclusive distribution of it. Matthew Jenusaitis, President of Boston
Scientific's Peripheral Interventions business, states, "The NexStent carotid
stent, used in combination with the Boston Scientific FilterWire EZ™ embolic
protection system, offers patients a less-invasive treatment alternative to
the traditional surgical procedure known as a carotid endarterectomy."
(source: Boston Scientific Corporation / EndoTex Interventional
Systems, Inc.)
April
11, 2005
Abbott
Submits StarClose™ Vascular Closure Device
for FDA Approval
Abbott Vascular today announced the submission of a Premarket Approval application
to the U.S. Food and Drug Administration (FDA) for its StarClose™ Vascular
Closure System, a next–generation vessel closure system specifically designed
for the simple, quick and secure mechanical closure of femoral arteries following
catheterization procedures, such as angioplasty and stent placement. Abbott
Vascular President Robert B. Hance stated, "As a leader in vessel closure,
Abbott is pleased to announce this important step in the development of a next–generation
closure device for the U.S. market.... In our pivotal clinical study, StarClose
was shown to close femoral arteries securely and safely in a matter of seconds,
reducing the amount of time patients need to remain at bedrest following a
catheterization procedure."
(source: Abbott Laboratories)
April
6, 2005
Shedding
new light on coronary bypass surgery
Although this article is not about interventional procedures per se, it does
illustrate how changes in imaging technology can imporve patient outcomes --
and this new imaging is being performed by Peter Fitzgerald, MD, PhD of Stanford,
one of the pioneers of intravascular ultrasound and its use in the coronaries.
(source: Stanford University)
March
31, 2005
Former
Secretary of State Kissinger undergoes heart
procedure
Kissinger, who is almost 82, underwent angioplasty on Tuesday and is now "resting
comfortably at home," according to Myrna Manners of New York-Presbyterian
Hospital/Weill Cornell Medical Center in Manhattan.
(source: Newsday)
March
30, 2005
Hoag
Hospital Announces First U.S. Patient Enrolled
in ACT I Clinical Trial
The first patient in the U.S. to be enrolled in the clinical trial named “Carotid
Angioplasty and Stenting vs. Endarterectomy in Asymptomatic Subjects with Significant
Extracranial Carotid Occlusive Disease Trial” (ACT I) was treated today
at the Hoag Heart and Vascular Institute at Hoag Memorial Hospital Presbyterian
in Newport Beach, CA. ACT I is the first trial to study the potential benefits
of minimally invasive carotid stenting in patients who would normally undergo
surgery, carotid endarterectomy (CEA), to prevent stroke.
(source: Hoag Hospital)
March 25,
2005
Blue
Cross And Blue Shield Association Study Shows
Consumers Want Information To Help Them Make
Healthcare Treatment Decisions
A RAND Corporation study for Blue Cross and Blue Shield shows that 70 percent
of consumers surveyed said they turn to the Internet most often to find the
information they need to make treatment decisions. About 60 percent said they
also go to their doctor. "This is a clear indication we should empower
consumers to become better informed when making critical healthcare decisions,” said
Maureen Sullivan of Blue Cross/Blue Shield. “The challenge for physicians,
hospitals and payers is to provide information to consumers in an easy-to-use
format that helps them understand their treatment choices."
(source: Blue Cross and Blue Shield)
March
24, 2005
Boston
Scientific Announces Jury Verdict in Patent Litigation
with Johnson & Johnson
The company's press release discusses the jury verdict validating one of several
claims that Johnson & Johnson said infringed on its Palmaz patent (the
other claims were not found to infringe). The product involved was the NIR® stent,
which was designed and manufactured by Medinol Ltd. and formerly sold by Boston
Scientific. It is an older product and no longer sold. A news article from
Reuters, titled "US
jury rules against Boston Scientific on patent" discusses the situation
futher.
(source: Boston Scientific Corporation / Reuters)
March 17,
2005
Boston
Scientific disputes article on Medinol
The company disputes the information in an article (below) from the Israeli
newspaper Yediot Ahronot and company spokesperson Paul Donovan stated, "We
have had no discussions with Medinol for quite some time regarding settlement
of our disputes, and we have made no settlement offers of any kind or any amount."
(source: Reuters)
Boston
Scientific offers Medinol $600m compensation
The “Yediot Ahronot” Hebrew daily reports that negotiations to
settle the bitter dispute between Israel stent manufacturer Medinol, owned
by Kobi and Judith Richter, and US company Boston Scientific, Medinol’s
former partner, have reached an advanced stage.
(source: Golan Hazani - "Yediot Ahronot")
March 15, 2005
Boston
Scientific Receives Two FDA Approvals on Cutting
Balloon™ Product Line
Press release from the company announced that the FDA has granted approvals
to the company for its Quincy Massachusetts Distribution Center as a sterilization
site and its Letterkenny, Ireland facility for manufacturing of the Cutting
Balloon product line.
(source: Boston Scientific Corporation)
March 11,
2005
Boston
Scientific Facility Receives 2005 Shingo Prize
for Excellence in Manufacturing
The Shingo Prize, dubbed the "Nobel Prize for manufacturing" by BusinessWeek
magazine, was awarded to Boston Scientific for its achievements in lean manufacturing.
(source: Boston Scientific Corporation)
March
8, 2005
Vasomedical
Announces Positive PEECH Trial Results at American College
of Cardiology
(source: Vasomedical, Inc.)
The most recent study of the effects external counterpulsation therapy (ECP
or EECP) were presented today at the ACC. The PEECH™ (Prospective Evaluation
of EECP® in Congestive Heart Failure) study, while small (187 patients in 29
centers) showed benefits and the results "support the use of EECP therapy
as an adjunctive treatment for chronic stable heart failure patients",
accordingly to principal investigator Dr. Arthur Feldman of Jefferson Medical
College of Thomas Jefferson University, Philadelphia.
February 21, 2005
Doctors
in state try stent in artery
A story about a patient treated at Sparks Regional Medical Center in Fort Smith,
Arkansas with a carotid stent (the Rx Acculink and Rx Accunet by Guidant).
Gives a good picture of the procedure.
(source: Tracie Dungan, Arkansas Democrat Gazette)
February 18,
2005
Study
Suggests Non-Surgical Cryoplasty® Therapy
Promising Treatment For Blocked Leg Arteries
Positive results from patients treated at the Texas Heart Institute for Peripheral
Artery Disease (PAD) were presented at the International Congress on Endovascular
Intervention XVIII today. This new therapy uses nitrous oxide in a balloon
catheter inside of saline to cool the artery to -10°C before and during
balloon expansion. The results in leg arteries have been very good, significantly
reducing the need for the additional treatment of stent placement. An information
packet on cryoplasty for physicians and patients is available at cryoplasty.com.
(source: CryoVascular Systems, Inc.)
February
18, 2005
Jury
rules against Medtronic in stent patent
A jury in a Delaware court ruled that some Medtronic's stent designs infringed
on patents held by rival stent-maker Guidant. The ruling had to do with older
stents, according to Medtronic's
press release, they are the Microstent, GFX, GFXII, S540, S660, S7, Driver,
Bridge, and BeStent2 -- most these basic designs were inherited from Arterial
Vascular Engineering (AVE) when Medtronic purchased them in 1998.
(source: Reuters)
February 18, 2005
Info
Sought on Planned J&J-Guidant Merger
The Federal Trade Commission (FTC) has asked for more information from Johnson & Johnson
and Guidant on the two companies' planned merger. In a joint
statement, the companies indicated that the request was anticipated, adding
that they still expect to receive regulatory approval and close the acquisition
during the third quarter of this year. For a history of the merger talks, see
our special section.
(source: Associated Press)
February
16, 2005
Clopidogrel
plus Aspirin: Cost-Effective for a Year
A study published yesterday by Mark D. Schleinitz, MD, MS, of Brown University
and P.A. Heidenreich, MD, MS of Stanford showed that patients with high-risk
acute coronary syndromes (unstable angina) not only lived slightly longer when
given a combination of clopidogrel (brand name: Plavix) and aspirin for the
first year after a cardiac event, as opposed to aspirin alone, but that this
treatment was "cost-effective", even when taking into account the
increased cost of clopidogrel.
(source: Annals of Internal Medicine)
February
16, 2005
Coil
Embolization Treatment 'Safe and Effective,'
According to British Health Authority Advisory
Committee
Demonstrating yet again the genius of Charles Dotter in conceiving of the circulatory
system as a "highway of the body", the Interventional Procedures
Advisory Committee (IPAC) of the United Kingdom's National Institute for Clinical
Excellence (NICE), has confirmed the safety and efficacy of coil embolization
for the treatment of ruptured and unruptured brain aneurysms. "Coil embolization
offers patients with a ruptured aneurysm a safer and less- invasive treatment
with an improved chance of recovery for a condition that could easily be fatal," said
Anil Gholkar, M.D., consultant neuroradiologist, of the Regional Neurosciences
Centre in Newcastle-upon-Tyne, England.
(source: Boston Scientific Corporation)
February
15, 2005
Guidant
Corp. Recalls Some Catheters
This voluntary recall involves balloons that are used to open clogged arteries,
specifically the Voyager Coronary Dilatation Catheters of sizes 1.5 mm to 3.5
mm (the Voyager RX was not part of the recall). The Voyager was launched
last July after F.D.A. approval. The reason given is a possible leak which
could allow air into the system. The company states in its filing, "While
the probability of a leak is low, the company is pursuing root cause analysis
and corrective actions."
(source: Associated Press)
February
14, 2005
Quitting
Smoking Improves Blood Tests in Days
A Japanese study found that 2 weeks after stopping smoking made the blood significantly
less likely to form clots (one of the major ways in which heart attacks occur).
This doesn't mean that the effects of smoking on the body are reversed in such
a short period, but it does show an immediate and very important benefit to
smoking cessation.
(source: Reuters)
February
7, 2005
Surgery
or angioplasty? New scale helps physicians choose best
heart treatment
In the current issue of the AHA's journal Circulation, Dr. John Spertus
of the Mid America Heart Institute of Saint Luke’s Hospital in Kansas
City, Missouri, reports the results of a study in which outcomes of patients
with varying levels of "restenosis risk" were rated. The report titled, "Risk
of Restenosis and Health Status Outcomes for Patients Undergoing Percutaneous
Coronary Intervention Versus Coronary Artery Bypass Graft Surgery" (read
the abstract here) showed that bypass surgery was better for patients with
a medium-to-high risk of restenosis (as rated using a scale developed for the
study). However, the study was based on patient data from 1999-2000 which pre-dates
the drug-eluting stent, a major new device which significantly reduces the
risk of restenosis. The authors, recognizing the impact of this new modality,
conclude that the risk of restenosis "...should also be tested as
a means for considering drug-eluting as opposed to bare metal stents in PCI." For
a further discussion of this study, also see our Editor's
Blog.
(source: American Heart Association)
February
7, 2005
Study
Shows "Time to Treatment" for Heart
Attack Using Angioplasty in the U.S. Takes Too
Long
This study used data from the National Registry of Myocardial Infarction to
see how quickly the transfer of heart attack patients to 419 hospitals that
perform angioplasty was being done in the "real world". Previous
studies have shown that angioplasty (or "primary PCI" -- Percutaneous
Coronary Intervention) has clear benefits for treatment of myocardial infarction
(see our Feature
Topic) if done within 90 minutes, currently the national guideline.
But this study reveals that the "door-to-balloon" time (the period
covering the patient arriving at the first hospital, diagnosed, transferred
to the second, brought into the cath lab and a balloon inflated) was within
90 minutes in less than 5% of patients, with a mean time of double that. Angioplasty
is still theoretically the best treatment but, as the authors conclude, "For
the full benefits of primary PCI to be realized in transfer patients, improved
systems are urgently needed to minimize total door-to-balloon times."
(source: Harlan M. Krumholz MD et al, Circulation)
February 7,
2005
Gum
Disease Seems to Clog Arteries
A study, published in this week's Circulation,
show a direct relationship between high levels of periodontal bacteria that
cause gum disease and early stages of atherosclerosis. The study used ultrasound
measurements of the arterial wall in carotid (neck) arteries.
(source: E.J. Mundell, HealthDay)
February 7,
2005
Abbott
Enters Into Agreement with Cleveland Clinic for
Myeloperoxidase (MPO) Cardiac Test
MPO's link to inflammation in coronary arteries from unstable plaque could
make it a potential biomarker useful for risk stratification in patients presenting
with chest pain. "Today, cardiac diagnostic tests already influence how
treatment recommendations are made. The potential of an MPO test to help identify
patients at risk for a major cardiac event in the near term could prove to
be another advance in patient care," said Stanley Hazen, M.D., Ph.D.,
head of the Section of Preventive Cardiology and Cardiac Rehabilitation at
The Cleveland Clinic.
(source: Abbott Laboratories)
February 1, 2005
Zylon
Corporation awarded major grant from NIH
The National Heart Lung and Blood Institute of the National Institutes of Health
has awarded a research grant to further develop Zylon's innovative "Ultra
low profile coronary stent delivery catheter".
(source: Zylon Corporation)
January 27, 2005
Abbott
sets trial of carotid stents
Another story about Abbott's carotid stenting trial, comparing the less invasive
endovascular approach to conventional "open" surgery.
(source: Bruce Japsen, Chicago Tribune)
January 19, 2005
St.
Jude Medical Ships 5 Millionth Angio-Seal Vascular
Closure Device
In the company's press release Paul R. Buckman, President of St. Jude Medical's
Cardiology Division states: "St. Jude Medical's Angio-Seal™ device is
now chosen by more physicians than any other vascular closure technology....
Many healthcare providers have concluded that the use of vascular closure devices
has become an indispensable part of the catheterization procedure. The shipment
of the 5 millionth Angio-Seal™ device signifies the market's overwhelming
acceptance of this technology. It is truly becoming the standard of care for
obtaining hemostasis."
(source: St. Jude Medical, Inc.)
January
18, 2005
Abbott
Announces Groundbreaking New Trial in Stroke
Prevention
The company announced today that it will begin enrolling patients in its recently
FDA-conditionally-approved clinical study, called the Asymptomatic Carotid
Trial or ACT I. It will be a multicenter, randomized trial that will compare
carotid artery stenting to carotid artery surgery in asymptomatic patients
(those who have not displayed symptoms of a stroke but are at–risk) who
normally would be referred for surgery. This is the first such major trial
of its type. Another
article from Reuters about the trial quotes Dr. Takao Ohki, chief of vascular
surgery at Montefiore Hospital in New York: "From an industry standpoint,
it has big opportunity. From a clinician and patient standpoint, it would allow
us to have more options in terms of what kind of treatment we could offer." You
can see and hear Dr. Ohki and many other vascular surgeons who are pioneering
the endovascular option in our documentary, "Vascular
Pioneers: Evolution of a Specialty".
(source: Abbott Laboratories / Reuters Health / VascularTherapy.Org)
January
18, 2005
Absorbable
Metal Stent Opens Blocked Leg Arteries
Initial results with a bio-absorbable metal stent show that the novel device
works as intended -- to prop open blocked leg arteries before being fully absorbed
into the artery wall, so that complications common with standard stents don't
occur.
(source: Megan Rauscher, Reuters)
January 18, 2005
Experimental
Stent Could Revolutionize Treatment of Artery
Disease
More on the bio-absorbable stent study that was presented at the annual
ISET meeting in Miami Beach.
(source: Ed Edelson, HealthDay)
January 18, 2005
Reinsdorf
OK after angioplasty
White Sox and Bulls owner Jerry Reinsdorf underwent artery-clearing angioplasty
Monday after routine tests found minor blockage, the White Sox said.
(source: NorthWest Herald) |
| Click
here for more information about the following ads
|
|
